Abstract
Purpose This study retrospectively investigated whether infarction in specific Alberta Stroke Program Early CT Score (ASPECTS) regions is associated with clinical outcome in patients with symptomatic non-acute internal carotid or middle cerebral artery occlusion who underwent endovascular recanalisation (ER).
Methods Preoperative ASPECTS and region of infarction were recorded before recanalisation. Clinical outcome was evaluated 90 days after the procedure using the modified Rankin Scale; a score>2 was defined as poor outcome. Secondary outcomes included postprocedural cerebral oedema, intracranial haemorrhage (ICH) and symptomatic ICH.
Results Among the 86 patients included, 90-day outcome was poor in 30 (34.9%) and 40 experienced cerebral oedema (46.5%). Multivariate logistic regression models showed that lenticular nucleus infarction (OR 19.61–26.00, p<0.05), admission diastolic blood pressure (OR 1.07–1.08, p<0.05), preprocedural National Institutes of Health Stroke Scale (OR 1.96–2.05, p<0.001) and haemorrhagic transformation (OR 14.99–18.81, p<0.05) were independent predictors of poor 90-day outcome. The area under the receiver operating characteristic curve for lenticular nucleus infarction as a predictor of poor outcome was 0.73. M2 region infarction (OR 26.07, p<0.001) and low American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology collateral circulation grade (OR 0.16, p=0.001) were independent predictors of postprocedural cerebral oedema. The area under the receiver operating characteristic curve for M2 region infarction as a predictor of cerebral oedema was 0.64. Region of infarction did not significantly differ between patients with and without postprocedural ICH or symptomatic ICH.
Conclusions Lenticular nucleus and M2 region infarction were independent predictors of poor 90-day outcome and postprocedural cerebral oedema, respectively, in patients with non-acute anterior circulation large artery occlusion who underwent ER.
WHAT IS ALREADY KNOWN ON THIS TOPIC
The 10 scoring regions of the anterior circulation Alberta Stroke Program Early CT Score (ASPECTS) are weighted differently for functional prognosis in patients treated with mechanical or intravenous thrombolysis. However, whether region is as important a predictor of clinical outcome as size or ASPECTS Score in patients who had a non-acute large vessel occlusion ischaemic stroke treated endovascular recanalisation (ER) remains unknown.
WHAT THIS STUDY ADDS
Lenticular nucleus and M2 region infarction were independent predictors of poor 90-day outcome and postprocedural cerebral oedema, respectively, in patients with non-acute anterior circulation large artery occlusion who underwent ER.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
Patients with infarct involving the lentiform nucleus and M2 region may need to undergo better preoperative evaluation and develop a more cautious surgical approach.
Introduction
Large artery occlusion is a major cause of ischaemic stroke. In a population-based study of 75 patients with symptomatic internal carotid artery (ICA) occlusion, the risk of stroke within 30 days, 1 year and 5 years of diagnosis was 0%, 8% and 15%, respectively.1
Endovascular recanalisation (ER) is a viable treatment option in patients with non-acute intracranial artery occlusion (defined as >24 hours between discovery of the arterial occlusion and treatment) and phase II cerebral haemodynamic failure who experience stroke recurrence despite standard pharmacological treatment and lifestyle management.2 The reported technical success rate of ER is as high as 90% and >80% of patients achieve a modified Rankin Scale (mRS) Score≤2 at the 90-day follow-up.2–4 The reported short-term risk of stroke or death associated with ER ranges between 8.5% and 19.4%; the reported long-term risk ranges from 4.7% to 11.7%.2–4 However, randomised controlled trials investigating criteria for ER in patients with non-acute large artery occlusion are lacking.
Most clinical studies of ER in non-acute large artery occlusion have excluded patients with an anterior circulation Alberta Stroke Program Early CT Score (ASPECTS)≤6.5–7 However, SELECT2 (Randomized Controlled Trial to Optimize Patient's Selection for Endovascular Treatment in Acute lschemic Stroke), ANGEL-ASPECT (Randomized Controlled Trial of Endovascular Therapy in Acute Anterior Circulation Large Vessel Occlusive Patients with a Large Infarct Core), and LASTE (LArge Stroke Therapy Evaluation trial) studies published in the past 2 years have shown that acute large artery occlusion patients with large infarcts (ASPECTS≤5) can also benefit from ER.8–10 The 10 scoring regions of the anterior circulation ASPECTS are weighted differently for functional prognosis and intracranial haemorrhage (ICH) in acute large artery occlusion patients treated with mechanical or intravenous thrombolysis.11–13
Furthermore, the association between region of infarction and clinical outcome in patients with symptomatic non-acute anterior circulation large artery occlusion undergoing ER has not been well studied. It is possible that region is as important a predictor of outcome as size or ASPECTS Score. Therefore, an ASPECTS of 6 or lower may not be an absolute exclusion criterion for ER of non-acute large artery occlusion. This study aimed to investigate the association of region of infarction with functional outcome and complications of ER in patients with non-acute anterior circulation large artery occlusion. Hopefully, our findings will provide fellow clinicians a reference to assist with treatment decision-making in these patients.
Materials and methods
Patients
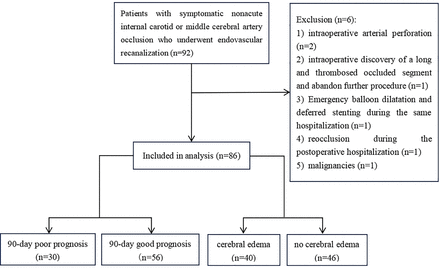
Patients with symptomatic non-acute anterior circulation large artery occlusion, treated using ER from January 2019 to January 2024 at the stroke centre of the research institution, were consecutive reviewed. Inclusion criteria were as follows: (1) ICA or middle cerebral artery (MCA) occlusion detected on magnetic resonance or CT angiography and confirmed by digital subtraction angiography; (2) intracranial atherosclerosis was the primary aetiology; (3) >24 hours between discovery of the arterial occlusion and treatment using ER; (4) A qualifying ischaemic stroke in the territory of the occluded ICA or MCA must have occurred within the past 12 months; (5) recurrent transient ischaemic attacks or stroke related to the occluded ICA or MCA despite optimal medical treatment that including antiplatelet therapy, statin, blood pressure and glucose control and an emphasis on a healthy lifestyle; and (6) postprocedural modified Thrombolysis in Cerebral Infarction Score≥2b. Patients with cancer and those who experienced intraoperative arterial perforation and ICH or were discovered to have a long and thrombosed occluded segment requiring abandonment of the procedure were excluded. We also excluded patients who underwent emergency balloon dilatation and deferred stenting during the same hospitalisation and those who developed reocclusion after the ER procedure.
Data collection
Demographic data, vascular risk factors, stroke details, laboratory data and surgical procedures were recorded by two trained researchers. Imaging data were independently reviewed by a neurointerventionalist and a neuroradiologist who were blinded to the clinical data. Any disagreements were resolved by consensus. ASPECTS was determined based on the most recent noncontrast CT (NCCT) or MRI examination performed in the 7 days prior to ER. In the lentiform nucleus, caudate nucleus, internal capsule and insula regions, 1 point was scored for the presence of non-punctate hypoattenuating lesions. In the cortical regions (M1–6), 1 point was scored for the presence of hypoattenuating lesions that comprised >30% of the regional area on NCCT or >20% on MRI.14 The technical approach in the procedures was discussed and decided on a case-by-case basis by a team of supervising physicians. All patients underwent brain NCCT 4 and 24 hours after ER.
Clinical outcomes
The mRS Score 90 days after ER was considered the primary outcome and was evaluated by two blinded investigators via outpatient or telephone interview; a score of 3–6 was defined as poor outcome. Secondary outcomes included postprocedural cerebral oedema, ICH and symptomatic ICH (sICH). Cerebral oedema was defined as loss of grey matter–white matter differentiation or widening of the cerebral gyrus and narrowing of the cerebral sulcus on NCCT. ICH was classified using the Heidelberg Bleeding Classification.15 sICH was defined as ≥4-point worsening of National Institutes of Health Stroke Scale (NIHSS) Score in conjunction with presence of ICH on NCCT in an area corresponding to the symptoms.15
Patient and public involvement
During follow-up, patients first participate in the research through outpatient review or telephone follow-up, and the results are evaluated by trained investigators based on a uniform mRS Questionnaire. Patients were not involved in the design, recruitment or conduct of the study. The clinical outcomes were collected during the normal course of medical care, so patients were not asked to assess the burden of the intervention and the time required to participate in the research. The patient disseminates the results of the research in a voluntary form, with no limitation on how and when.
Statistical analysis
Statistical analyses were performed using SPSS software V.26.0 (IBM, Armonk, New York, USA). Continuous variables are presented as medians with IQR or means with SD as appropriate and were compared using the independent samples t-test or Mann-Whitney U test. Categorical variables are presented as frequencies with percentage and were compared using the χ2 test or Fisher’s exact test. The variance inflation factor was used to analyse multicollinearity between ASPECTS and the regions of infarction; a factor>10 indicates severe covariance between variables. Variables with multicollinearity were manually excluded.16 The degree of correlation between regions of infarction was analysed using Spearman’s method: r>0.5 was considered a strong correlation.17 Variables with p<0.05 in the univariate comparisons between the good and poor outcome groups and the clinically recognised influencing factors were included in the multivariate logistic regression analysis. The stepwise withdrawal method was used to improve multivariate test efficacy when the number of outcome events was small and the number of relevant variables in the univariate analysis was large. p<0.05 was considered significant.
Results
Patient characteristics
A study flowchart is shown in figure 1. Among the 86 patients who met criteria and were included for analysis, 65 were men (75.6%). The median age was 61 years (range, 54–66). Median preprocedural NIHSS Score was 4 (range, 1–7). Median ASPECTS Score before ER was 8 (range, 7–9). Median American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology (ASITN/SIR) collateral circulation score was 2 (range, 2–3). Median time between discovery of the arterial occlusion and ER was 17 days (range, 9–28). Laterality of occluded artery was left in 42 patients (48.8%). The occlusion was intracranial in 49 patients (57.0%).
Flowchart for screening research subjects.
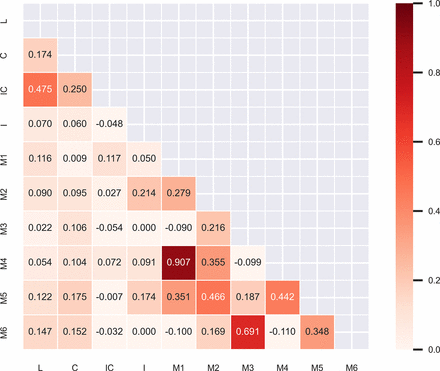
Infarction was most frequent in the lenticular nucleus (49 patients (57.0%)), followed by the insula (43 patients (50.0%)). The M1 region (5 patients (5.8%)) was least frequent. Infarction of the M1 region was strongly correlated with infarction of the M4 region (r=0.907). A strong correlation was also found for coinfarction of the M3 and M6 regions (r=0.691). Correlation coefficients for coinfarction in the lenticular nucleus and internal capsule (r=0.475) and the M2 and M5 regions (r=0.466) did not meet the criteria for strong correlation, but was significant. A correlation heat map is shown in figure 2.
Correlation coefficients heat map of Alberta Stroke Program Early CT Score regions. Infarction of the M1 region was strongly correlated with infarction of the M4 region (r=0.907). A strong correlation was also found for coinfarction of the M3 and M6 regions (r=0.691). Correlation coefficients for coinfarction in the lenticular nucleus and internal capsule (r=0.475) and the M2 and M5 regions (r=0.466) did not meet the criteria for strong correlation, but was significant.
90-day clinical outcomes
Clinical outcome at 90 days was poor in 30 patients (34.9%). Patient characteristics in the good and poor outcome groups are shown in table 1. The frequency of infarction involving the lenticular nucleus (86.7% vs 41.1%, p<0.001), M2 region (30.0% vs 10.7%, p=0.025) and M5 region (26.7% vs 5.4%, p=0.013) was significantly higher in the poor outcome group. In addition, median preprocedural NIHSS Score (9 vs 2), mean diastolic blood pressure at admission (90.1 vs 83.0 mm Hg) and haemorrhagic transformation (56.7% vs 17.9%) were significantly higher and preprocedural ASPECTS was significantly lower (7 vs 8) in the poor outcome group. The other characteristics did not significantly differ between groups. Because of covariance between ASPECTS and infarct regions in variance inflation factor analysis, ASPECTS was not included in the multivariate logistic regression models (online supplemental tables 5 and 6).
Supplementary data
Baseline characteristics between the 90-day poor outcome and good outcome groups
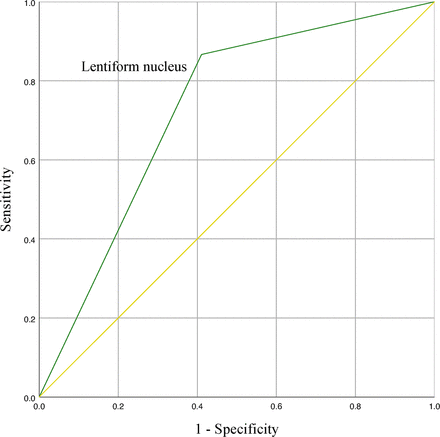
The results of the multivariate logistic regression analyses are shown in table 2. All the models indicated that lenticular nucleus infarction, DBP on admission, preprocedural NIHSS Score and haemorrhagic transformation were independent predictors of poor 90-day outcome. Receiver operating characteristic analysis was performed to assess the value of lenticular nucleus infarction as a predictor of poor outcome, which found an area under the curve of 0.73 (95% CI, 0.62 to 0.84, p=0.001) with sensitivity and specificity of 87% and 59%, respectively (figure 3).
Multivariate logistic regression models of 90-day poor outcome
Receiver operating characteristic curve with lenticular nucleus infarcts. The area under the curve of lenticular nucleus infarction as a predictor of poor outcome was 0.73 (95% CI, 0.62 to 0.84, p=0.001) with sensitivity and specificity of 87% and 59%, respectively.
Secondary outcomes
Postprocedural cerebral oedema was present in 40 patients (46.5%). Frequency of M2 region infarction (32.5% vs 4.3%) and median preprocedural NIHSS Score (5 vs 3) were significantly higher and ASITN/SIR Score (2 vs 2) was significantly lower in patients with cerebral oedema than those without. The two groups did not significantly differ in terms of the other characteristics (table 3). In multivariate logistic regression analysis, M2 region infarction (OR 26.07; 95% CI, 4.57 o 148.54; p<0.001) and low ASITN/SIR Score (OR 0.16; 95% CI, 0.06 to 0.47; p=0.001) were independent predictors of cerebral oedema (table 4). In receiver operating characteristic analysis of the predictive value of M2 region infarction for postoperative cerebral oedema, the area under the curve was 0.64 (95% CI, 0.52 to 0.76; p=0.025) with sensitivity and specificity of 33% and 96%, respectively (figure 4).
Receiver operating characteristic curve with M2 region infarcts. In receiver operating characteristic analysis of the predictive value of M2 region infarction for postoperative cerebral oedema, the area under the curve was 0.64 (95% CI, 0.52 to 0.76; p=0.025) with sensitivity and specificity of 33% and 96%, respectively.
Baseline characteristics of patients with or without cerebral oedema
Multivariate logistic regression model of postoperative cerebral oedema
Region of infarction did not significantly differ between patients with and without ICH or sICH after ER (online supplemental tables 7 and 8).
Discussion
In our study of patients with symptomatic non-acute anterior circulation large artery occlusion treated with ER, lenticular nucleus infarction significantly increased the risk of poor 90-day outcome while M2 region infarction significantly increased the risk of postprocedural cerebral oedema. Several previous studies have found an association between region of infarction and clinical outcome in patients treated with mechanical thrombectomy or intravenous thrombolysis. A previous large retrospective study of patients who had an anterior circulation stroke from the Albumin in Acute Stroke trials database found that lenticular nucleus infarction was independently associated with poor outcome in those who underwent intravenous thrombolysis; when the infarction involved the lenticular and caudate nuclei, insula and M5 region, the risk of sICH was increased by a factor of 1.62.12 Even a study in which poor outcome was defined as mRS Score>1, which includes those with even slight disability, reported that lenticular nucleus infarction was an independent predictor of poor outcome.18 Considering the subjective differences between individuals in assessing ASPECTS, Ni et al used RAPID software (iSchemaView) to automatically analyse the location of the ischaemic core on CT perfusion images before thrombolysis and found that the risk of ICH increased sevenfold when the infarction involved the subcortical region; moreover, the risk of sICH increased as the volume of the subcortical region infarction increased.19 Sato et al reported that upper limb motor impairment items of the NIHSS Score were higher (more impaired) when small subcortical infarcts involved the lenticular nucleus, corona radiata and internal capsule.20 Compared with other neurological deficits in stroke, upper limb impairment usually lasts longer.21 In addition, the lenticular nucleus has also been associated with language22 and swallowing function,23 both of which significantly affect quality of life. A meta-analytic connectivity modelling study found that the left basal ganglia was more strongly associated with poor outcome than the right, presumably owing to the greater impact of the dominant hand on daily life.22 However, whether lentiform nucleus infarction is associated with clinical outcome after ER remains controversial, as some studies have reported it is not.24 25
The association between lentiform nucleus infarction and poor outcome can be explained by neuroanatomy and regional connectivity. The lentiform nucleus, which comprises the putamen and globus pallidus, is involved in control of somatic voluntary movement and connects the cerebral cortex to the basal ganglia.26 Signals from the motor cortex are relayed to the putamen, processed and transmitted to the globus pallidus27; most of the signals are then returned to the cortex via the ventrolateral nucleus of the thalamus, forming a corticobasal ganglionic loop.28 Damage to the lentiform nucleus results in disruption of this loop. Depending on the specific site of infarction, both orofacial and limb motor function may be affected.26 The putamen, lateral segment of the globus pallidus, head of the caudate nucleus, internal capsule and corona radiata adjacent to the internal capsule are supplied by the lenticulostriate arteries; therefore, infarction involving the dorsolateral aspect of the lentiform nucleus may also damage the caudate nucleus, internal capsule and the corona radiata, resulting in extensive damage to the corticobasal ganglionic loops which are associated with limb motor function.29 Our correlation heat map also demonstrates this, showing a significant correlation between the lentiform nucleus and internal capsule. There may also be an association between the lentiform nucleus and the caudate nucleus.17
M2 region infarction was a risk factor for cerebral oedema after non-acute ER in our study. Although the association between region of infarction and cerebral oedema has not been previously examined, several studies have reported the association between infarction region and postprocedural ICH or sICH. Their conclusions are contrary. In a study of anterior circulation arterial occlusion patients with ASPECTS<6 treated using mechanical thrombolysis, Xing et al found that the lower the ASPECTS in the cortical region, the higher the risk of postoperative ICH and malignant cerebral oedema.30 Gu et al examined 108 patients who had an acute stroke treated using intravenous thrombolysis and found that cortical ASPECTS was independently associated with both ICH (OR 0.197) and sICH (OR 0.066); in addition, cortical ASPECTS had a higher predictive accuracy for haemorrhagic transformation than ASPECTS.31 Several studies have shown that infarction in the M2 and M5 regions is significantly associated with poor functional outcome when ASPECTS is assessed using NCCT or CT perfusion images.11 13 However, others have suggested that neither the M2 region nor other cortical regions are significantly associated with functional outcome or postprocedural ICH or sICH.24 25
Collateral small vessel hypoplasia and blood–brain barrier disruption are possible reasons why M2 region infarction increases the risk of postprocedural cerebral oedema. After re-establishing blood flow with recanalisation of an occluded artery, the newly developing collateral small vessels in cortical regions are more prone to localised disruption of endothelial cell tight junctions, which leads to blood–brain barrier damage.32 When anterior circulation artery occlusion occurs, the blood supply to subcortical regions can be compensated by the circle of Willis; supply to the cortex is usually compensated by the leptomeningeal collateral vessels.32 The immature structure of the endothelium of the new leptomeningeal collateral vessels can result in increased permeability of the blood–brain barrier in the cortical region.33 If ER is performed at this point, the contrast agent and mechanical stimulation from the guidewire and balloon may aggravate tight junction dysfunction and endothelial structure destruction, resulting in complete disruption of the blood–brain barrier and impaired cerebrovascular autoregulation.34 A rapid increase in cerebral perfusion after revascularisation when autoregulation is impaired and extravasation of macromolecules through the damaged blood–brain barrier can cause cerebral oedema and even ICH.35 Tan et al reported a close association between collateral circulation status in the M5 region and functional outcomes in patients treated using intravenous thrombolysis.36 Our study and another16 both found that the M2 region is significantly associated with the M5 region. Therefore, we speculate that the status of collateral circulation in the M2 region has a potential impact on clinical outcome in patients with non-acute occlusion undergoing ER.
Permeability surface (PS) measured on CT perfusion imaging is an indicator of blood–brain barrier permeability, with elevated values representing barrier disruption. Li et al found that elevated relative PS in the MCA territory was the best predictor of ICH after ER of acute large vessel occlusion.37 In another study of patients who had an acute stroke with MCA occlusion, the risk of ICH was over 15 times higher in those with both poor collateral circulation and high relative PS.38
Our study showed that NIHSS Score, DBP at admission and haemorrhagic transformation were independently associated with poor 90-day outcome and that low ASITN/SIR Score was independently associated with postprocedural cerebral oedema, which are consistent with the findings of previous studies.13 39 M5 region infarction is a possible predictor of poor 90-day outcome, as shown in our study and several previous ones, with the possible rationale as described above.11 13
Due to the small sample size, this study cannot reach the conclusion that ASPECTS<6 are not the absolute exclusion criteria for ER of non-acute large artery occlusion. According to the latest expert consensus in China, the infarction area exceeding 1/2 blood vessel supply area is still the exclusion criteria for chronic occlusion recanalisation treatment.40 However, the results of LASTE, ANGEL-ASPECT and SELECT2 studies showed that ASPECTS<6 are not the absolute exclusion criteria for acute large artery occlusion recirculation treatment.8–10 Previous clinical experience of our centre also found that some patients with non-acute symptomatic large artery occlusion could still benefit from revascularisation treatment, even if ASPECTS<6. This retrospective study verified this experience. Among the 10 patients who had preoperative ASPECTS<6, 4 patients had postoperative ICH (the overall incidence was 31.4%) and 3 cases had good outcome at 90 days (the overall incidence was 65.1%). It is expected that future large-scale prospective studies will further explore this topic.
Our study has several limitations. First, confounding may have been present because of its single-centre retrospective design, even though we adjusted for many factors. Second, we could not accurately analyse the effect of region of infarction on postoperative ICH or sICH because of its small sample size. Third, the small number of infarctions in certain regions affected the accuracy of our statistical analyses.
Conclusion
Lenticular nucleus and M2 region infarction were independent predictors of poor 90-day outcome and postprocedural cerebral oedema, respectively, in patients with non-acute anterior circulation large artery occlusion treated using ER. Future large multicentre prospective studies are required to determine the effect of infarction location on clinical outcome.
Data availability statement
Data are available on reasonable request. The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics statements
Patient consent for publication
Ethics approval
This study involves human participants and was approved by the ethics committee of the Second Affiliated Hospital of Xi’an Jiaotong University (No. 2021008). Participants gave informed consent to participate in the study before taking part.
Acknowledgments
We thank Liwen Bianji (Edanz) (https://www.liwenbianji.cn) for editing the English text of a draft of this manuscript.
Footnotes
Contributors Lu Jialiang and Zhang Guilian is responsible for the overall content [as guarantor]. All authors contributed to the conception and methodology of the work and the acquisition of data. Zhang Guilian contributed to the validation, writing (review and editing), supervision, project administration, resources and funding acquisition. Lu Jialiang contributed to the formal analysis, data curation, writing (original draft preparation, review and editing) and visualization. All authors approved the final version of the manuscript to be published.
Funding This work was supported by the National Natural Science Foundation of China (grant no. 82471333) and the National Natural Science Foundation of China (grant no. 81971116).
Competing interests None declared.
Provenance and peer review Not commissioned; externally peer reviewed.
Supplemental material This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
This is an open access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited, appropriate credit is given, any changes made indicated, and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/.